METAL BIOAVAILABILITY IN SURFACE WATERS
Description:
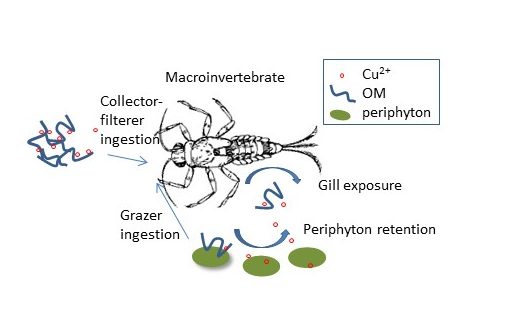
This project deals with quantifying the dynamics of organic carbon-metal interactions and exposure and how that influences trophic transfer. A subset of contaminant metals, e.g. Cu and Zn, are essential elements for biological function but are also common toxicological concerns in urban water bodies. This project will help with understanding the protectiveness of the present regulations against these metals that are based only on dissolved metal concentrations in developed freshwater streams.
The goal of this project is to identify metal speciation and bioavailability predictors of biological uptake in primary producers and higher trophic level organisms of different feeding classes and the dependence on metal loading dynamics.
Graduate Students: Nafis Fuad, Katie Turpin-Nagel, Hongwei Luan,
Funding Sources: NSF CAREER, CT IWR
Publications:
Turpin-Nagel, K. & Vadas, T.M. (2016). Controls on metal exposure to aquatic organisms in urban streams. Environmental Science Processes & Impacts. Vol 18, pp. 956 – 967.
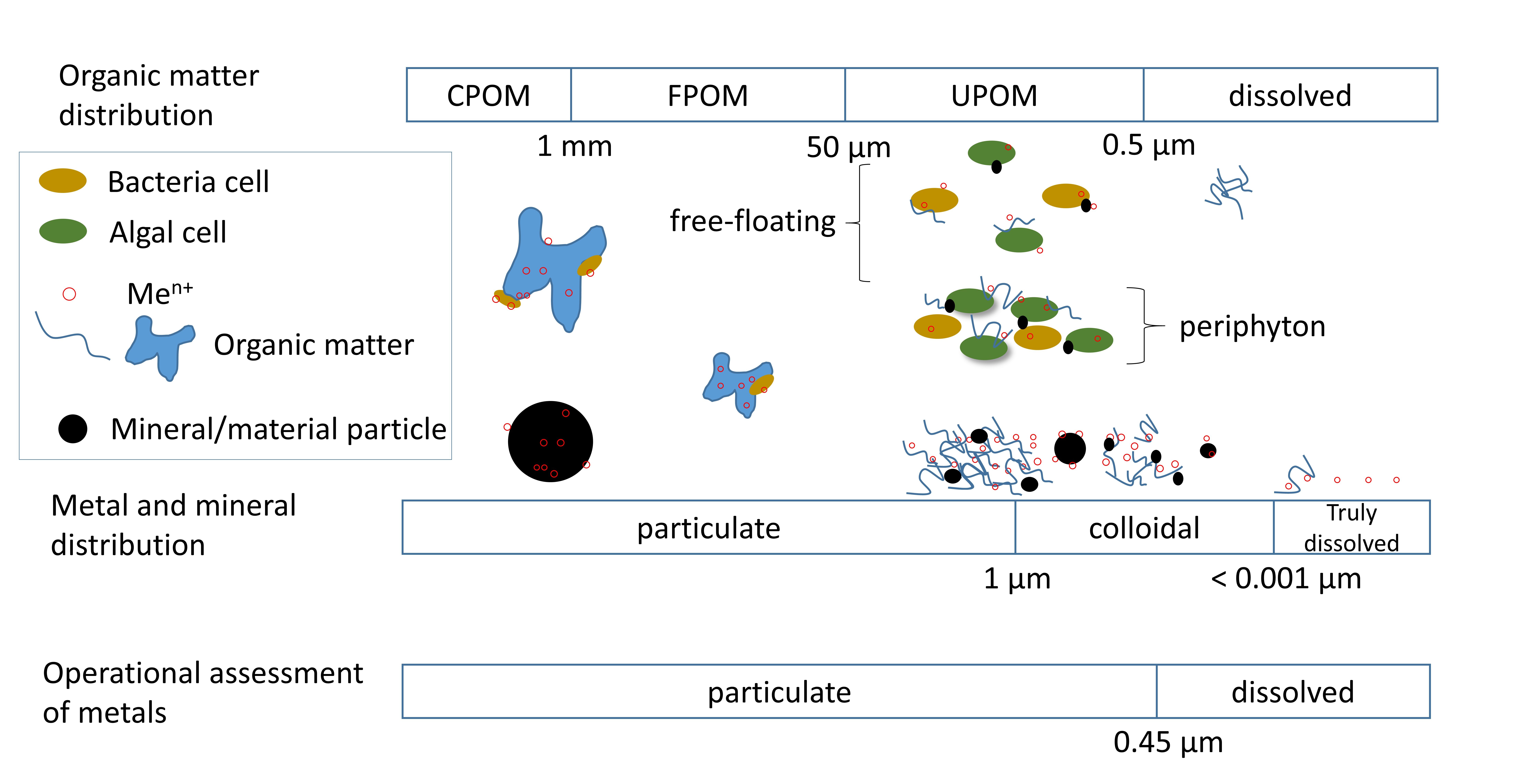
Luan, H. & Vadas, T.M. (2015) Size characterization of dissolved metals and organic matter in source waters to streams in developed landscapes. Environmental Pollution. 197:76-83.
Periphyton Exposure Experiments
BIOGEOCHEMISTRY IN WETLAND SYSTEMS
Description:
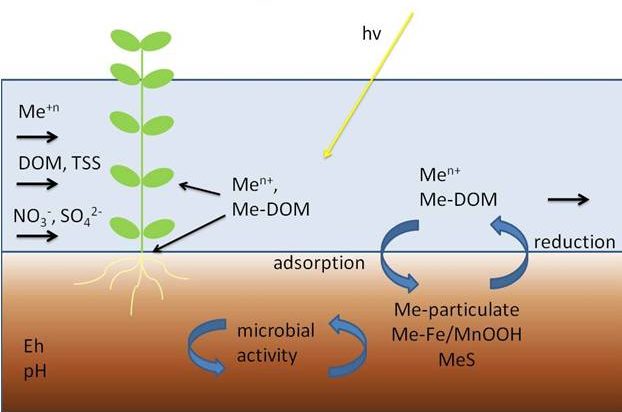
The unique processes seen in wetlands have led to many studies on their functions and the implementation of constructed wetlands such as treatment wetlands. Constructed wetlands are commonly used in urban areas or areas where runoff and stormwater accumulate and flooding and/or treatment of stormwater is of concern. Wetlands receive inconsistent flows which create fluctuations between wet and dry, or anoxic and oxic conditions. During these shifts in redox potential, common wetland elemental cycles such as iron (Fe) reduction, oxidation, and hydrolysis, organic matter (OM) turnover, and trace metal retention are altered. This can lead to variability in OM and metal retention.
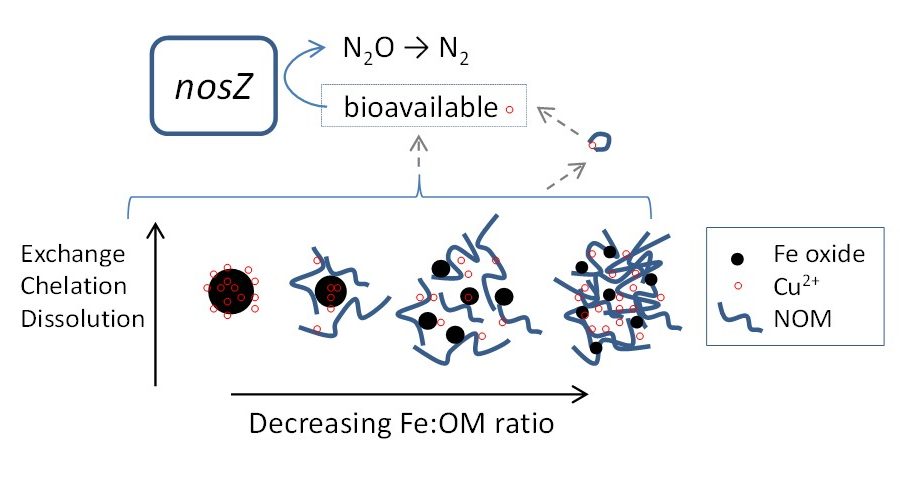
While natural wetlands are abundant in OM and trace metals, our understanding of how these constructed systems function may be skewed due to the exposure of different input loads. Constructed wetlands receive inputs that typically contain higher nutrient, metal, and OM concentrations from anthropological sources. Therefore, processes such as Fe cycling, metal retention, and availability may be altered in constructed wetlands. Observations in treatment wetlands have also shown Cu interacting with OM and Fe oxides as it encounters soil and surface water zones high in organic matter (OM) and amid Fe cycling. The increased presence of OM during Fe cycling results in higher OM and Cu uptake via formation of ternary complexes in the bulk phase and possibly a lowered expected availability within the dissolved phase as components form ternary complexes in the colloidal phases. This all suggests that within treatments wetlands, Cu and OM availability may be limited thereby negatively impacting processes such as denitrification which require both.
Graduate Students: Randi Mendes, Neila Seda
Funding Sources: USGS, CT IWR, CT Sea Grant
Publications:
Seda, N., Koenigsmark, F., & Vadas, T.M. (2016). Sorption and coprecipitation of copper to ferrihydrite and humic acid organomineral complexes and controls on copper availability. Chemosphere. 147:272-278.
Aqueous Phase Coprecipitation Reaction
WATER REUSE
Description: Disinfection byproducts (DBPs) are compounds that form unintentionally during chlorination for pathogen control within the water distribution network, typically due to a reaction with organic matter (OM). There are over 500 identified DBPs, including iodinated, brominated, and chlorinated compounds. Some DBPs are known carcinogens, but very few are regulated by the EPA. The formation conditions of DBPs depend on pH, reaction time, Br- or I- present, type of oxidant, and other water chemistry parameters. Additionally, in water reuse applications of different source waters, varying natural organic matter (NOM) concentrations and compositions could lead to the increased formation of more toxic DBPs.
Graduate Students: Julia Czarnecki
Funding Sources: USDA
Publications:
PLANT UPTAKE OF CONTAMINANTS
Description:
Exploring alternative water sources is one potential strategy for overcoming agricultural water scarcity issues. When considering new sources, it is important to address the presence of potential contaminants, including pharmaceutical compounds (PCs) and disinfection byproducts (DBPs). Ongoing experiments are investigating these compounds and their uptake in experimental applications of treated wastewater reuse in hydroponic greenhouse grown lettuce, Lactuca sativa.
Graduate Students: Julia Czarnecki, Dorottya Kelemen
Funding Sources: USDA
Publications:

CARBON NANOMATERIALS FOR WATER TREATMENT
Description:
Fabricated the new form of activated carbon as nonwoven nano-structure membrane based on precursor polymer- polyacrylonitrile and composites of a membrane with carbon nanotube and tested the optimal conditions for membrane by controlling stabilization, carbonization and activation procedures, as well as the effect of additives, such as phthalic acid, etc. These carbonaceous materials have been modified by different chemical pathways to introduce different functional groups, targeting specific micropollutants. These novel membrane material has been applied into adsorption by comparing with commercial activated carbon materials.
Graduate Students: Yi Han
Funding Sources:
Publications:
Han, Y. R. Li, C. Brückner, T.M. Vadas. (2018). Controlling the surface oxygen groups of polyacrylonitrile-based carbon nanofiber membranes while limiting fiber degradation. Membranes' C. 4 (3) 40.